Index > GREATER BAY AREA INTERNATIONAL CLINICAL TRIAL INSTITUTE
Turning Research into Real-World Impact
Context
The Greater Bay Area International Clinical Trial Institute (GBAICTI) is a one-stop platform that coordinates clinical trial resources across Hong Kong’s public and private sectors, including universities, the Hospital Authority, and private healthcare facilities.
GBAICTI is a government-backed, academically operated institute designed not just to conduct research, but to genuinely transform the conditions under which research happens.
We were tasked with creating a visual identity that could communicate GBAICTI’s unique proposition and its industry context.



The Guangdong-Hong Kong-Macao Greater Bay Area is one of the most ambitious regional development projects in the world. Linking two Special Administrative Regions, Hong Kong and Macao, and nine cities in Guangdong Province, it is home to over 86 million people and an economy that rivals those of some of the world’s largest nations.
What makes the GBA genuinely interesting for life sciences is its structural complexity. Three separate legal systems operate within the same geography. That is both its biggest challenge and, when navigated well, its biggest advantage. Hong Kong brings international regulatory credibility; Shenzhen and the mainland bring scale, infrastructure, and a vast patient population. The Shenzhen-Hong Kong-Guangzhou cluster ranks second globally for patents and publications, and the GBA accounts for over 50% of China's Phase III clinical trial registrations.
The earliest recorded controlled experiment in medicine dates to 1061 in China, in the Ben Cao Tu Jing (Atlas of Materia Medica). Two men were asked to run together; only one was given ginseng beforehand. The untreated runner reportedly struggled for breath, while the other remained steady.
This is widely regarded as one of the earliest recorded examples of a control group: comparing someone who receives a treatment with someone who does not. That same principle still underpins modern clinical trials today.
Image credits: 1. Wellcome Collection; 2. James Lind Library
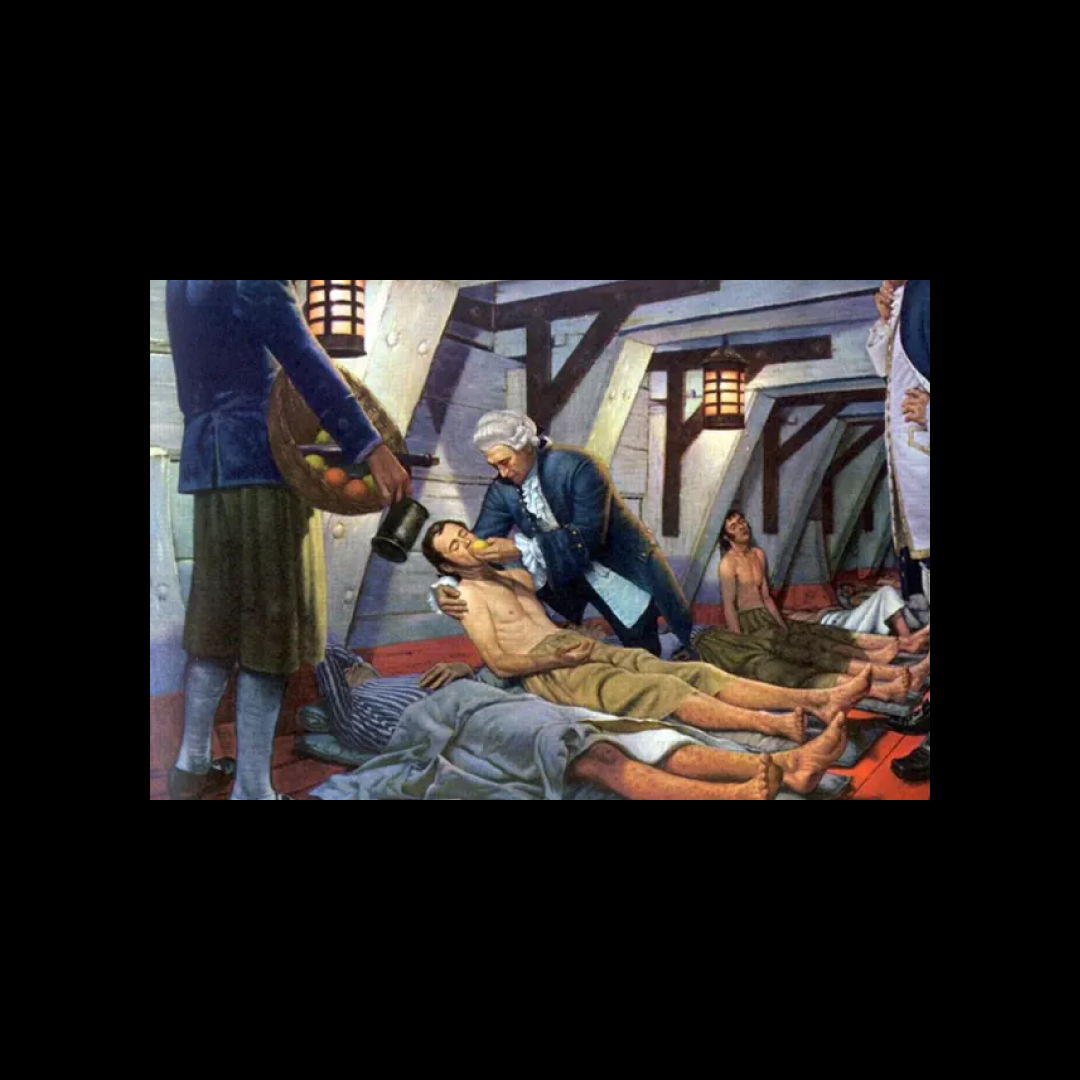
Nearly seven hundred years later, that same logic played out aboard a Royal Navy vessel. In 1747, Scottish physician James Lind was trying to understand scurvy, which was killing sailors at a devastating rate.
He selected twelve men, divided them into pairs, and gave each pair a different supposed remedy: cider, vinegar, seawater, citrus fruits, and others.
The sailors given citrus recovered most clearly. It was not a perfect study by modern standards, but it was methodical, comparative, and consequential. A lemon tree still features on the crest of the Institute of Naval Medicine in recognition of his work.
Image credits: 1. Robert Alan, James Lind, Conqueror of Scurvy, 1959; 2. George Chalmers, Portrait of James Lind, 1783. Private collection, Hepner family; 3. James Lind Library; 4. Institute of Naval Medicine, Royal Navy badge.





The global clinical trials market is projected to reach over $100 billion by 2030.¹
Asia Pacific is the fastest-growing region, and China is increasingly at the centre of that momentum. In 2024, 4,900 clinical trials were registered in China, a 13.9% increase on the previous year, with cell and gene therapy registrations growing by 42% in a single year.²
Sources:
1. MarketsandMarkets, Clinical Trial Services Market Size, Growth, Share & Trends Analysis, Report Code PH 1622, March 2025.
2. China’s Centre for Drug Evaluation (CDE), Annual Report on the Progress of Clinical Trials for New Drug Registration in China (2024), National Medical Products Administration, June 2025. Via Covington & Burling LLP, Lexology, 21 July 2025.

Yet for all its scale, the industry has a persistent blind spot. Its visual language defaults to data and laboratories. The human side, the patients, the anxiety, and the hope are largely absent.
Direction
We spent time with the GBAICTI team through workshops and conversations with researchers, administrators, clinicians, and government stakeholders who brought the institute into being. We wanted to understand not just what GBAICTI does, but what it believes, and where it wants to go.
What came through clearly was an organisation occupying genuinely rare ground: internationally credible, institutionally rigorous, yet in the early stages of building a public identity. The expertise and ambition were already there. What was needed was a unified language for it, something that could speak to a pharmaceutical partner in New York as naturally as it could to a researcher in Shenzhen or a patient in Guangzhou.
Four values emerged from those conversations.

Core Value 1
Integrity & Good Clinical Practice
Clinical research is only valuable if it is trustworthy, and trust is built through the consistent application of internationally recognised standards. Good Clinical Practice is the foundation on which every partner relationship, every delivered outcome, and every assurance to a participant rests. The same standards are applied to every study, at every site, without exception.
Core Value 2
Connection & Collaboration
The GBA’s greatest asset is its density. Within a compact geography, you have world-class universities, one of Asia’s most advanced hospital networks, a deep manufacturing base, and direct access to international markets. GBAICTI exists to activate that density, to bridge the fragmentation that has historically limited what the region could offer as a research environment, and to connect it with global networks. Collaboration here is structural, not aspirational.
Core Value 3
Transformation & Trust
The landscape is shifting fast: digital trials, AI-assisted analysis, compressed timelines. The institute has been designed to move with that change rather than resist it. But transformation without trust creates instability. The two have to move together. As processes modernise and the ecosystem becomes more agile, the consistency that participants and partners depend on must remain constant.
Core Value 4
Impact & Innovation
The measure of any clinical research institution is whether its work reaches patients. Discovery that stays in the laboratory, or research that never translates into accessible treatment, falls short. GBAICTI’s impact is measured in therapies approved, lives improved, and the region's growing contribution to global medical progress. Innovation is the engine; impact is the point.

Identity
Built from the Chinese 人 character, the logomark places people at the heart of our identity, reflecting our human-centric mission in clinical research. The individuals who participate in clinical trials, the communities they serve, and the professionals who advance medical discovery.

The clinical research industry consistently frames its work as something that happens to patients rather than for them. Participants are frequently invisible in the visual language of the institutions conducting research on their behalf.
GBAICTI is built on a human-centred philosophy, and 人 makes that explicit from the very first mark.


The logo echoes a dandelion releasing its seeds: a point of origin, and from it, ideas and discoveries carried outward into the world. It is an image that resonates naturally in the context of clinical research, the careful cultivation of knowledge, and its deliberate dispersal to the places where it can matter.
Flora held specific significance in the earliest recorded clinical experiments in China, the ginseng root, documented in Song Su's Ben Cao Tu Jing, was the subject of what may be the world's first recorded controlled trial. The botanical quality of the logomark carries that lineage forward quietly, without being heavy-handed about it.


The circular network maps directly onto GBAICTI's structure: GBA at the centre, connected to Guangdong, and outward to global research partners.
It symbolises GBAICTI’s role in building a cross-border ecosystem where collaboration, standards, and innovation flow freely.

The colour palette was created to convey positivity and energy within the clinical context.
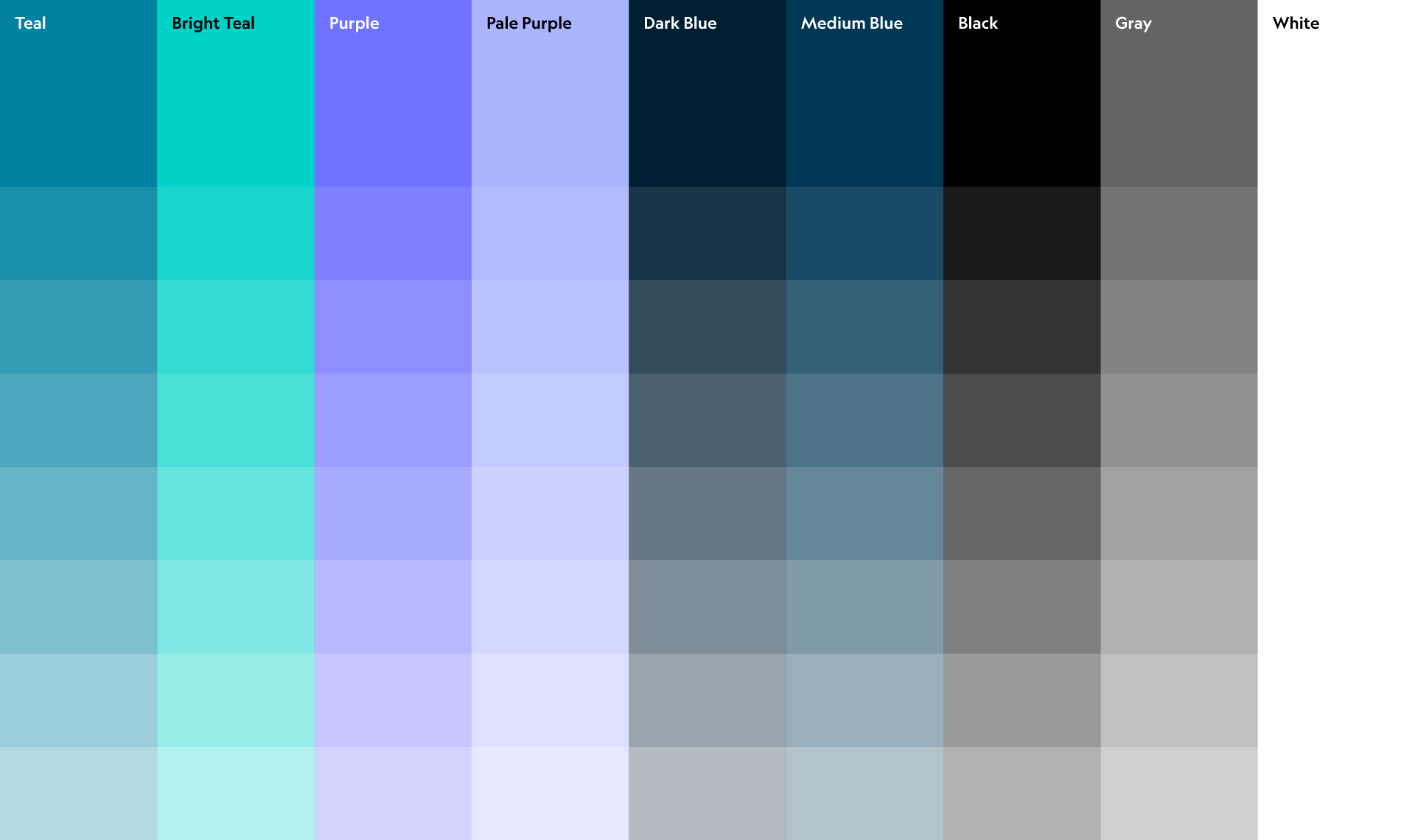
We used three weights of Geograph, a contemporary and geometric sans-serif typeface from Klim Foundry.
Geograph gives the brand identity system a confident, legible voice.

Alongside the logomark, we developed icons to support GBAICTI’s communications across digital and print. Each icon maps directly to one of the institute’s core values or service areas, providing the system with a functional layer that extends beyond the brand mark itself.
Research & Data Support
— Product Safety Assurance
Connection & Collaboration
Integrity & Good Clinical Practice
Impact & Innovation
Transformation & Trust
Community & Talent Cultivation
System-wide Process Optimization
Expression
















